Our Aim is to provide a Continuous Supply of High Tech Sophisticated and market shortage Pharmaceutical Products for the Middle East and North Africa Region through Egypt and Gulf offices.
Business Development
Driving revenue through
strategic partnership building
OUR MISSION
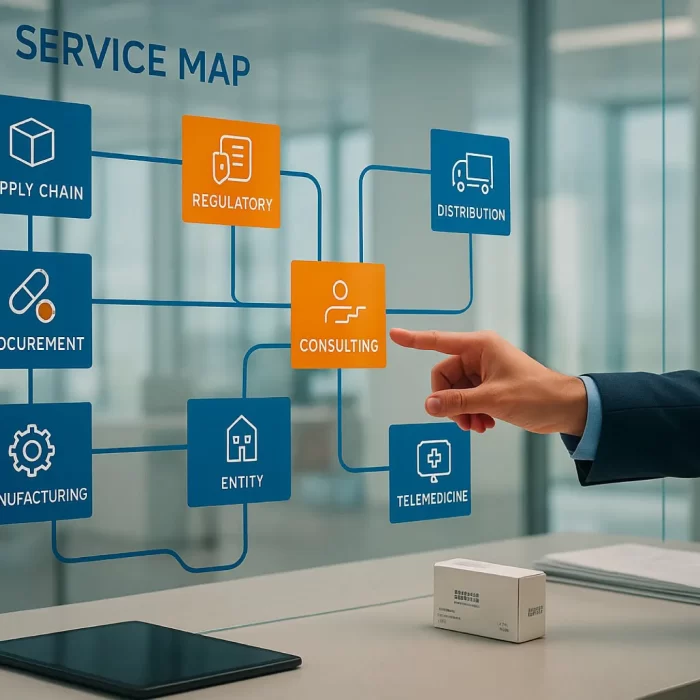
Selling Agenting
Our vision is to be recognized as one of the biggest importers of Science Led Innovation and Hi Tech Quality products across the Pharmaceutical Field, so that Future Generations can explore and enjoy healthy life
OUR VISION
Selling Agenting
Our patient safety is at the heart of everything we do. Our pharmacovigilance (PV) services are designed to ensure that our pharmaceutical products are continually monitored for safety, efficacy, and quality throughout their lifecycle. We are committed to maintaining the highest standards of safety, in compliance with global regulatory requirements.
Our Services Include:
1. Adverse Event Reporting and Management
- We provide a comprehensive system for the collection, analysis, and reporting of adverse drug reactions (ADRs) to health local authorities in each country we are operating in .
- Our team ensures that every incident is thoroughly investigated and documented in compliance with regulatory guidelines.
2. Signal Detection and Risk Management
Our risk management strategies help mitigate potential risks associated with our products, ensuring patient safety.
3. Regulatory Compliance and Reporting
We ensure adherence to all regulatory requirements, including periodic safety update reports (PSURs), risk evaluation and mitigation strategies (REMS), and expedited reporting for serious adverse events (SAEs). Our experienced team ensures timely and accurate submissions to regulatory agencies.
4. Pharmacovigilance Audits and Inspections
We conduct internal audits and assist in external inspections to assess compliance with pharmacovigilance regulations. Our services ensure that all processes meet the stringent standards set by regulatory authorities.
5. Training and Consultation
Our experts provide training to internal teams on pharmacovigilance best practices and emerging safety concerns.
6. Post-Market Surveillance and Data Analytics
We gather real-world data from various sources, including patient registries, and healthcare databases, to monitor product safety. Through data analytics, we identify trends, detect signals, and proactively manage safety concerns.
SEPARATE ENTITLES,
UNIQUE HARMONY
ONE MISSION
Mission Pharma Head Office
Building No. 6, Misr Aviation Association Division, next to Al Watany Eye Hospital, El Thawra Street, Heliopolis, Misr Al-Gedida – Cairo – Egypt
Mission Pharma Secondary Office
Mountain View ICity business park - new capital.
International Pioneers office (Mission Pharma affiliate)
3225, Ibn hubbira Street, Al Zahra District, Riyadh city, Saudi Arabia.
International Pioneers office 2 (Mission Pharma affiliate)
3171, Jabal Abi Naqba ST, AL Masani District, Riyadh City, Saudi Arabia.
-
Home
01
Home -
Who We Are
02
Who We Are -
Discover the power of one
03
Discover the power of one -
Services
04
Services -
Products
05
Products -
Innovation Access & Protection
07
Innovation Access & Protection -
Mission Sync
08
Mission Sync -
Career
06
Career -
Contact Us
09
Contact Us