Building No. 6, Misr Aviation Association Division, next to Al Watany Eye Hospital, El Thawra Street, Heliopolis, Misr Al-Gedida – Cairo – Egypt
Services
- Home
- Innovation Access & Protection
Innovation Access & Protection

1.
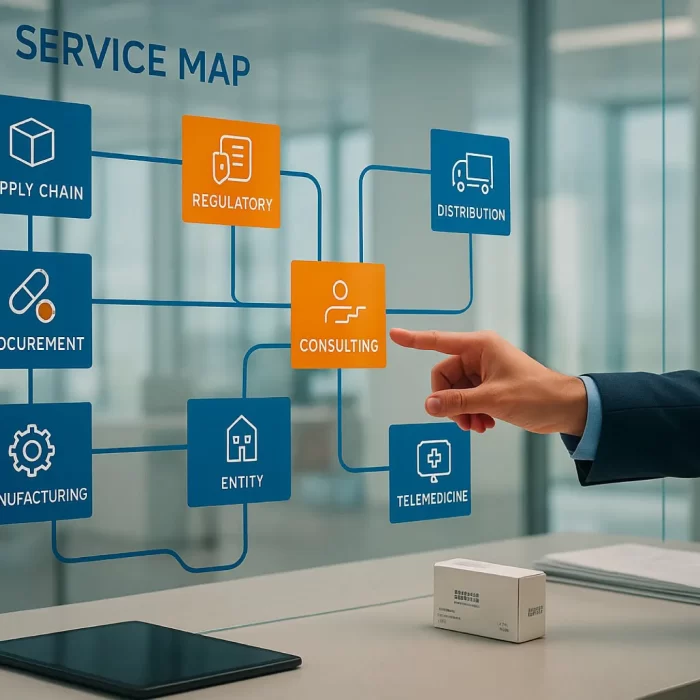
Scientific Office & Regulatory Representation
Our company operates a fully established Scientific Office in Saudi Arabia, compliant with Saudi Food & Drug Authority (SFDA) and GCC regulatory requirements, enabling seamless regulatory, medical, and scientific representation for patented and innovative products.
Our Scientific Office Services Include:
Full regulatory liaison with SFDA and regional health authorities
Product registration and lifecycle management for innovative medicines
Scientific communication and medical information services
Support for variations, renewals, and post-approval changes
Local Qualified Person support and regulatory compliance oversight
Management of regulatory documentation and authority correspondence
Our Scientific Office ensures your patented products are represented by a locally licensed, compliant, and scientifically competent team, reducing regulatory risk and accelerating market access.
2.
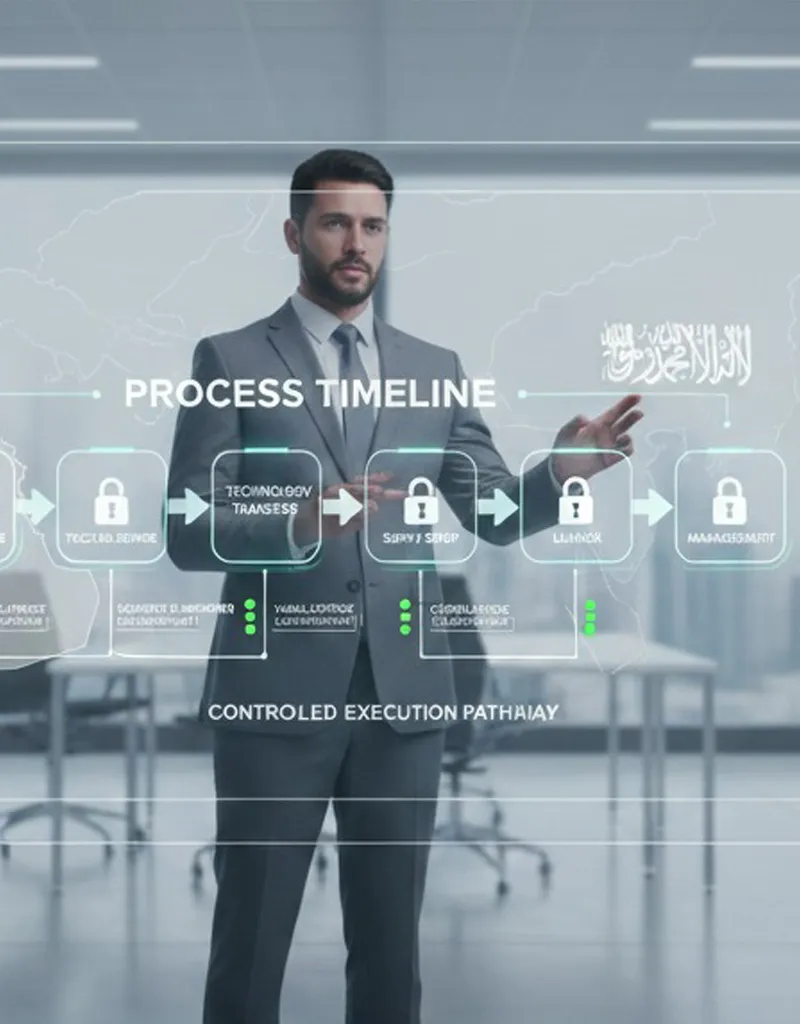
Structured Workflow & SOP-Driven Execution
We operate under a comprehensive set of Standard Operating Procedures (SOPs) that define every operational step from contract signature through commercialization and post-launch management.
Our Workflow Includes:
Confidentiality Agreement (CDA/NDA) execution
Due diligence and technical documentation review
Technology transfer and data package evaluation
Regulatory strategy alignment and submission planning
Product registration and authority interactions
Supply chain setup and serialization integration
Commercial launch planning and market access execution
Ongoing lifecycle management and performance reporting
Our SOP-Driven Approach Ensures:
Full traceability
Regulatory compliance
Timely execution
Transparency for the innovator partner
This structured workflow protects your asset and ensures consistent execution across Saudi Arabia, GCC, and MENA.

3.
Pharmacovigilance (PV) Agreement & QPPV Support
We maintain a fully operational Pharmacovigilance system and are prepared to sign a dedicated Pharmacovigilance Agreement in alignment with SFDA, GVP, and international pharmacovigilance requirements.
Our PV Capabilities Include:
Appointed and qualified local QPPV
Adverse Event (AE) and Safety Case processing
Local literature monitoring
Risk Management Plan (RMP) implementation
Safety data exchange agreements
Periodic Safety Update Reports (PSUR/PBRER) coordination
Authority inspections readiness
Our PV infrastructure ensures continuous patient safety monitoring and full compliance with local and global pharmacovigilance obligations for innovative and patented products.
4.
GDP & Supply Chain Excellence
We don’t just move products; we protect your innovation. Our distribution network is built on the rigorous standards of Good Distribution Practice (GDP), ensuring every life-saving treatment maintains its integrity from the moment it enters the country until it reaches the patient.
Why Partners Trust Our Supply Chain:
Certified Facilities: Modern, SFDA-compliant warehouses designed for pharmaceutical safety.
Climate Integrity: End-to-end temperature-controlled storage and logistics (Cold Chain & Ambient).
Real-Time Vigilance: Continuous 24/7 thermal monitoring and digital data logging.
Total Transparency: Full serialization ensures every unit is accounted for and traceable.
Elite Logistics: We utilize only validated transport lanes and highly vetted logistics partners.
Proactive Compliance: Sophisticated recall and complaint management systems for total accountability.
At the heart of our supply chain is a commitment to the patients who rely on your medicines. By adhering to the highest GDP standards, we ensure that every product remains safe, secure, and effective throughout its journey.


5.
Our IP & Patent Protection Fortress
Your intellectual property is the lifeblood of your company. We treat your patents with the same rigor we apply to the medicines themselves—providing a secure, legally fortified environment where innovation is respected and defended.
How We Protect Your Intellectual Property:
Operational Integrity: Custom SOPs designed to keep your technical data private.
Legal Scaffolding: Robust Non-Disclosure (NDA) and Non-Compete safeguards built into every contract.
Access Control: Strict limitations on who can view proprietary data.
Integrity Controls and Market Boundaries: Strict anti-diversion and anti-parallel trade controls to protect your territory.
Global Compliance: Fully aligned with both local SFDA regulations and international IP laws.
Our commitment to Intellectual Property (IP) goes beyond compliance—it is a core pillar of our business ethics, ensuring your assets are commercialized without compromise.
Mission Pharma Head Office
Mission Pharma Secondary Office
Mountain View ICity business park - new capital.
International Pioneers office (Mission Pharma affiliate)
3225, Ibn hubbira Street, Al Zahra District, Riyadh city, Saudi Arabia.
International Pioneers office 2 (Mission Pharma affiliate)
3171, Jabal Abi Naqba ST, AL Masani District, Riyadh City, Saudi Arabia.
-
Home
01
Home -
Who We Are
02
Who We Are -
Discover the power of one
03
Discover the power of one -
Services
04
Services -
Products
05
Products -
Innovation Access & Protection
07
Innovation Access & Protection -
Mission Sync
08
Mission Sync -
Career
06
Career -
Contact Us
09
Contact Us